- Latest available (Revised)
- Original (As adopted by EU)
Commission Directive 2006/86/ECShow full title
Commission Directive 2006/86/EC of 24 October 2006 implementing Directive 2004/23/EC of the European Parliament and of the Council as regards traceability requirements, notification of serious adverse reactions and events and certain technical requirements for the coding, processing, preservation, storage and distribution of human tissues and cells (Text with EEA relevance)
You are here:
- Directives originating from the EU
- 2006 No. 86
- Annexes only
- Show Geographical Extent(e.g. England, Wales, Scotland and Northern Ireland)
- Show Timeline of Changes
More Resources
Revised version PDFs
- Revised 29/04/20150.31 MB
This is a legislation item that originated from the EU
After exit day there will be three versions of this legislation to consult for different purposes. The legislation.gov.uk version is the version that applies in the UK. The EU Version currently on EUR-lex is the version that currently applies in the EU i.e you may need this if you operate a business in the EU.
The web archive version is the official version of this legislation item as it stood on exit day before being published to legislation.gov.uk and any subsequent UK changes and effects applied. The web archive also captured associated case law and other language formats from EUR-Lex.
Status:
EU Directives are being published on this site to aid cross referencing from UK legislation. After IP completion day (31 December 2020 11pm) no further amendments will be applied to this version.
ANNEX IU.K.Requirements for accreditation, designation, authorisation or licensing of tissue establishments as referred to in Article 3
A.ORGANISATION AND MANAGEMENTU.K.
1.A responsible person must be appointed having qualifications and responsibilities as provided in Article 17 of Directive 2004/23/EC.U.K.
2.A tissue establishment must have an organisational structure and operational procedures appropriate to the activities for which accreditation/designation/authorisation/licensing is sought; there must be an organisational chart which clearly defines accountability and reporting relationships.U.K.
3.Every tissue establishment must have access to a nominated medical registered practitioner to advise on and oversee the establishment’s medical activities such as donor selection, review of clinical outcomes of applied tissues and cells or interaction as appropriate with clinical users.U.K.
4.There must be a documented quality management system applied to the activities for which accreditation/designation/authorisation or licensing is sought, in accordance with the standards laid down in this Directive.U.K.
5.It must be ensured that the risks inherent in the use and handling of biological material are identified and minimised, consistent with maintaining adequate quality and safety for the intended purpose of the tissues and cells. The risks include those relating in particular to the procedures, environment, staff health status specific to the tissue establishment.U.K.
6.Agreements between tissue establishments and third parties must comply with Article 24 of Directive 2004/23/EC. Third party agreements must specify the terms of the relationship and responsibilities as well as the protocols to be followed to meet the required performance specification.U.K.
7.There must be a documented system in place, supervised by the responsible person, for ratifying that tissues and/or cells meet appropriate specifications for safety and quality for release and for their distribution.U.K.
8.In the event of termination of activities the agreements concluded and the procedures adopted in accordance with Article 21(5) of Directive 2004/23/EC shall include traceability data and material concerning the quality and safety of cells and tissues.U.K.
9.There must be a documented system in place that ensures the identification of every unit of tissue or cells at all stages of the activities for which accreditation/designation/authorisation/licensing is sought.U.K.
B.PERSONNELU.K.
1.The personnel in tissue establishments must be available in sufficient number and be qualified for the tasks they perform. The competency of the personnel must be evaluated at appropriate intervals specified in the quality system.U.K.
2.All personnel should have clear, documented and up-to-date job descriptions. Their tasks, responsibilities and accountability must be clearly documented and understood.U.K.
3.Personnel must be provided with initial/basic training, updated training as required when procedures change or scientific knowledge develops and adequate opportunities for relevant professional development. The training programme must ensure and document that each individual:U.K.
has demonstrated competence in the performance of their designated tasks;
has an adequate knowledge and understanding of the scientific/technical processes and principles relevant to their designated tasks;
understands the organisational framework, quality system and health and safety rules of the establishment in which they work, and
is adequately informed of the broader ethical, legal and regulatory context of their work.
C.EQUIPMENT AND MATERIALSU.K.
1.All equipment and material must be designed and maintained to suit its intended purpose and must minimise any hazard to recipients and/or staff.U.K.
2.All critical equipment and technical devices must be identified and validated, regularly inspected and preventively maintained in accordance with the manufacturers' instructions. Where equipment or materials affect critical processing or storage parameters (e.g. temperature, pressure, particle counts, microbial contamination levels), they must be identified and must be the subject of appropriate monitoring, alerts, alarms and corrective action, as required, to detect malfunctions and defects and to ensure that the critical parameters are maintained within acceptable limits at all times. All equipment with a critical measuring function must be calibrated against a traceable standard if available.U.K.
3.New and repaired equipment must be tested when installed and must be validated before use. Test results must be documented.U.K.
4.Maintenance, servicing, cleaning, disinfection and sanitation of all critical equipment must be performed regularly and recorded accordingly.U.K.
5.Procedures for the operation of each piece of critical equipment, detailing the action to be taken in the event of malfunctions or failure, must be available.U.K.
6.The procedures for the activities for which accreditation/designation/authorisation/licensing is sought, must detail the specifications for all critical materials and reagents. In particular, specifications for additives (e.g. solutions) and packaging materials must be defined. Critical reagents and materials must meet documented requirements and specifications and when applicable the requirements of Council Directive 93/42/EEC of 14 June 1993 concerning medical devices(1) and Directive 98/79/EC of the European Parliament and of the Council of 27 October 1998 on in vitro diagnostic medical devices(2).U.K.
D.FACILITIES/PREMISESU.K.
1.A tissue establishment must have suitable facilities to carry out the activities for which accreditation/designation/authorisation or licensing is sought, in accordance with the standards laid down in this Directive.U.K.
2.When these activities include processing of tissues and cells while exposed to the environment, this must take place in an environment with specified air quality and cleanliness in order to minimise the risk of contamination, including cross-contamination between donations. The effectiveness of these measures must be validated and monitored.U.K.
3.Unless otherwise specified in point 4, where tissues or cells are exposed to the environment during processing, without a subsequent microbial inactivation process, an air quality with particle counts and microbial colony counts equivalent to those of Grade A as defined in the current European Guide to Good Manufacturing Practice (GMP), Annex 1 and Directive 2003/94/EC is required with a background environment appropriate for the processing of the tissue/cell concerned but at least equivalent to GMP Grade D in terms of particles and microbial counts.U.K.
4.A less stringent environment than specified in point 3 may be acceptable where:U.K.
a validated microbial inactivation or validated terminal sterilisation process is applied;
or, where it is demonstrated that exposure in a Grade A environment has a detrimental effect on the required properties of the tissue or cell concerned;
or, where it is demonstrated that the mode and route of application of the tissue or cell to the recipient implies a significantly lower risk of transmitting bacterial or fungal infection to the recipient than with cell and tissue transplantation;
or, where it is not technically possible to carry out the required process in a Grade A environment (for example, due to requirements for specific equipment in the processing area that is not fully compatible with Grade A).
5.In point 4(a), (b), (c) and (d), an environment must be specified. It must be demonstrated and documented that the chosen environment achieves the quality and safety required, at least taking into account the intended purpose, mode of application and immune status of the recipient. Appropriate garments and equipment for personal protection and hygiene must be provided in each relevant department of the tissue establishment along with written hygiene and gowning instructions.U.K.
6.When the activities for which accreditation/designation/authorisation or licensing is sought involve storage of tissues and cells, the storage conditions necessary to maintain the required tissue and cell properties, including relevant parameters such as temperature, humidity or air quality must be defined.U.K.
7.Critical parameters (e.g. temperature, humidity, air quality) must be controlled, monitored, and recorded to demonstrate compliance with the specified storage conditions.U.K.
8.Storage facilities must be provided that clearly separate and distinguish tissues and cells prior to release/in quarantine from those that are released and from those that are rejected, in order to prevent mix-up and cross-contamination between them. Physically separate areas or storage devices or secured segregation within the device must be allocated in both quarantine and released storage locations for holding certain tissue and cells collected in compliance with special criteria.U.K.
9.The tissue establishment must have written policies and procedures for controlled access, cleaning and maintenance, waste disposal and for the re-provision of services in an emergency situation.U.K.
E.DOCUMENTATION AND RECORDSU.K.
1.There must be a system in place that results in clearly defined and effective documentation, correct records and registers and authorised Standard Operating Procedures (SOPs), for the activities for which accreditation/designation/authorisation/licensing is sought. Documents must be regularly reviewed and must conform to the standards laid down in this Directive. The system must ensure that work performed is standardised, and that all steps are traceable; i.e. coding, donor eligibility, procurement, processing, preservation, storage, transport, distribution or disposal, including aspects relating to quality control and quality assurance.U.K.
2.For every critical activity, the materials, equipment and personnel involved must be identified and documented.U.K.
3.In the tissue establishments all changes to documents must be reviewed, dated, approved, documented and implemented promptly by authorised personnel.U.K.
4.A document control procedure must be established to provide for the history of document reviews and changes and to ensure that only current versions of documents are in use.U.K.
5.Records must be shown to be reliable and a true representation of the results.U.K.
6.Records must be legible and indelible and may be handwritten or transferred to another validated system, such as a computer or microfilm.U.K.
7.Without prejudice to Article 9(2), all records, including raw data, which are critical to the safety and quality of the tissues and cells shall be kept so as to ensure access to these data for at least 10 years after expiry date, clinical use or disposal.U.K.
8.Records must meet the confidentiality requirements laid down in Article 14 of Directive 2004/23/EC. Access to registers and data must be restricted to persons authorised by the responsible person, and to the competent authority for the purpose of inspection and control measures.U.K.
F.QUALITY REVIEWU.K.
1.An audit system must be in place for the activities for which accreditation/designation/authorisation/licensing is sought. Trained and competent persons must conduct the audit in an independent way, at least every two years, in order to verify compliance with the approved protocols and the regulatory requirements. Findings and corrective actions must be documented.U.K.
2.Deviations from the required standards of quality and safety must lead to documented investigations, which include a decision on possible corrective and preventive actions. The fate of non-conforming tissues and cells must be decided in accordance with written procedures supervised by the responsible person and recorded. All affected tissues and cells must be identified and accounted for.U.K.
3.Corrective actions must be documented, initiated and completed in a timely and effective manner. Preventive and corrective actions should be assessed for effectiveness after implementation.U.K.
4.The tissue establishment should have processes in place for review of the performance of the quality management system to ensure continuous and systematic improvement.U.K.
ANNEX IIU.K.Requirements for the authorisation of tissue and cell preparation processes at the tissue establishments as referred to in Article 4
The competent authority shall authorise each tissue and cell preparation process after evaluation of the donor selection criteria and procurement procedures, the protocols for each step of the process, the quality management criteria, and the final quantitative and qualitative criteria for cells and tissues. This evaluation must comply at least with the requirements set out in this Annex.
A.RECEPTION AT THE TISSUE ESTABLISHMENTU.K.
Upon reception of procured tissues and cells at the tissue establishment, the tissues and cells must comply with the requirements defined in Directive 2006/17/EC.
B.PROCESSINGU.K.
When the activities for which the accreditation/designation/authorisation/licensing is sought include processing of tissues and cells, the tissue establishment procedures must comply with the following criteria:
The critical processing procedures must be validated and must not render the tissues or cells clinically ineffective or harmful to the recipient. This validation may be based on studies performed by the establishment itself, or on data from published studies or, for well established processing procedures, by retrospective evaluation of the clinical results for tissues supplied by the establishment.
It has to be demonstrated that the validated process can be carried out consistently and effectively in the tissue establishment environment by the staff.
The procedures must be documented in SOPs which must conform to the validated method and to the standards laid down in this Directive, accordingly with Annex I(E), points 1 to 4.
It must be ensured that all processes are conducted in accordance with the approved SOPs.
Where a microbial inactivation procedure is applied to the tissue or cells, it must be specified, documented, and validated.
Before implementing any significant change in processing, the modified process must be validated and documented.
The processing procedures must undergo regular critical evaluation to ensure that they continue to achieve the intended results.
Procedures for discarding tissue and cells must prevent the contamination of other donations and products, the processing environment or personnel. These procedures must comply with national regulations.
C.STORAGE AND RELEASE OF PRODUCTSU.K.
When the activities for which the accreditation/designation/authorisation/licensing is sought include storage and release of tissues and cells, the authorised tissue establishment procedures must comply with the following criteria:
Maximum storage time must be specified for each type of storage condition. The selected period must reflect among others possible deterioration of the required tissue and cell properties.
There must be a system of inventory hold for tissues and/or cells to ensure that they cannot be released until all requirements laid down in this Directive have been satisfied. There must be a standard operating procedure that details the circumstances, responsibilities and procedures for the release of tissues and cells for distribution.
A system for identification of tissues and cells throughout any phase of processing in the tissue establishment must clearly distinguish released from non-released (quarantined) and discarded products.
Records must demonstrate that before tissues and cells are released all appropriate specifications are met, in particular all current declaration forms, relevant medical records, processing records and test results have been verified according to a written procedure by a person authorised for this task by the responsible person as specified in Article 17 of Directive 2004/23/EC. If a computer is used to release results from the laboratory, an audit trail should indicate who was responsible for their release.
A documented risk assessment approved by the responsible person as defined in Article 17 of Directive 2004/23/EC must be undertaken to determine the fate of all stored tissues and cells following the introduction of any new donor selection or testing criterion or any significantly modified processing step that enhances safety or quality.
D.DISTRIBUTION AND RECALLU.K.
When the activities for which the accreditation/designation/authorisation/licensing is sought include distribution of tissues and cells, the authorised tissue establishment procedures must comply with the following criteria:
Critical transport conditions, such as temperature and time limit must be defined to maintain the required tissue and cell properties.
The container/package must be secure and ensure that the tissue and cells are maintained in the specified conditions. All containers and packages need to be validated as fit for purpose.
Where distribution is carried out by a contracted third party, a documented agreement must be in place to ensure that the required conditions are maintained.
There must be personnel authorised within the tissue establishment to assess the need for recall and to initiate and coordinate the necessary actions.
An effective recall procedure must be in place, including a description of the responsibilities and actions to be taken. This must include notification to the competent authority.
Actions must be taken within pre-defined periods of time and must include tracing all relevant tissues and cells and, where applicable, must include trace-back. The purpose of the investigation is to identify any donor who might have contributed to causing the reaction in the recipient and to retrieve available tissues and cells from that donor, as well as to notify consignees and recipients of tissues and cells procured from the same donor in the event that they might have been put at risk.
Procedures must be in place for the handling of requests for tissues and cells. The rules for allocation of tissues and cells to certain patients or health care institutions must be documented and made available to these parties upon request.
A documented system must be in place for the handling of returned products including criteria for their acceptance into the inventory, if applicable.
E.FINAL LABELLING FOR DISTRIBUTIONU.K.
1.The primary tissue/cell container must provide:U.K.
type of tissues and cells, identification number or code of the tissue/cells, and lot or batch number where applicable;
identification of the tissue establishment;
expiry date;
in the case of autologous donation, this has to be specified (for autologous use only) and the donor/recipient has to be identified;
in the case of directed donations - the label must identify the intended recipient;
when tissues and cells are known to be positive for a relevant infectious disease marker, it must be marked as: BIOLOGICAL HAZARD[F1;]
[F2Single European Code as applicable to the tissues and cells being distributed for human application or the donation identification sequence as applicable to the tissues and cells released for circulation, other than distributed for human application.]
Textual Amendments
[F1If any of the information under points (d), (e) and (g) above cannot be included on the primary container label, it must be provided on a separate sheet accompanying the primary container. This sheet must be packaged with the primary container in a manner that ensures that they remain together.]
2.The following information must be provided either on the label or in accompanying documentation:U.K.
description (definition) and, if relevant, dimensions of the tissue or cell product;
morphology and functional data where relevant;
date of distribution of the tissue/cells;
biological determinations carried out on the donor and results;
storage recommendations;
instructions for opening the container, package, and any required manipulation/reconstitution;
expiry dates after opening/manipulation;
instructions for reporting serious adverse reactions and/or events as set out in Articles 5 to 6;
presence of potential harmful residues (e.g. antibiotics, ethylene oxide etc)[F1;]
[F2for imported tissues and cells, the country of procurement and the exporting country (if different from the procurement country).]
F.EXTERNAL LABELLING OF THE SHIPPING CONTAINERU.K.
For transport, the primary container must be placed in a shipping container that must be labelled with at least the following information:
identification of the originating tissue establishment, including an address and phone number;
identification of the organisation responsible for human application of destination, including address and phone number;
a statement that the package contains human tissue/cells and HANDLE WITH CARE;
where living cells are required for the function of the graft, such as stem cells gametes and embryos, the following must be added: ‘DO NOT IRRADIATE’;
recommended transport conditions (e.g. keep cool, in upright position, etc.);
safety instructions/method of cooling (when applicable).
[F1ANNEX III U.K. NOTIFICATION OF SERIOUS ADVERSE REACTIONS
PART A U.K. Rapid notification for suspected serious adverse reactions
Tissue establishment
EU tissue establishment code (if applicable)
Report identification
Reporting date (year/month/day)
Individual affected (recipient or donor)
Date and place of procurement or human application (year/month/day)
Unique donation identification number
Date of suspected serious adverse reaction (year/month/day)
Type of tissues and cells involved in the suspected serious adverse reaction
Single European Code of tissues or cells involved in the suspected serious adverse reaction (if applicable)
Type of suspected serious adverse reaction(s)
PART B U.K. Conclusions of Serious Adverse Reactions Investigation
Tissue establishment
EU tissue establishment code (if applicable)
Report identification
Confirmation date (year/month/day)
Date of serious adverse reaction (year/month/day)
Unique donation identification number
Confirmation of serious adverse reaction (Yes/No)
Single European Code of tissues or cells involved in the confirmed serious adverse reaction (if applicable)
Change of type of serious adverse reaction (Yes/No) If YES, specify
Clinical outcome (if known)
Complete recovery
Minor sequelae
Serious sequelae
Death
Outcome of the investigation and final conclusions
Recommendations for preventive and corrective actions
ANNEX IV U.K. NOTIFICATION OF SERIOUS ADVERSE EVENTS
PART A U.K. Rapid notification for suspected serious adverse events
| Tissue establishment | ||||
| EU tissue establishment code (if applicable) | ||||
| Report identification | ||||
| Reporting date (year/month/day) | ||||
| Date of serious adverse event (year/month/day) | ||||
| Serious adverse event, which may affect quality and safety of tissues and cells due to a deviation in: | Specification | |||
|---|---|---|---|---|
| Tissues and cells defect | Equipment failure | Human error | Other (specify) | |
| Procurement | ||||
| Testing | ||||
| Transport | ||||
| Processing | ||||
| Storage | ||||
| Distribution | ||||
| Materials | ||||
| Others (specify) | ||||
PART B U.K. Conclusions of Serious Adverse Events investigation
Tissue establishment
EU tissue establishment code (if applicable)
Report identification
Confirmation date (year/month/day)
Date of serious adverse event (year/month/day)
Root cause analysis (details)
Corrective measures taken (details)]
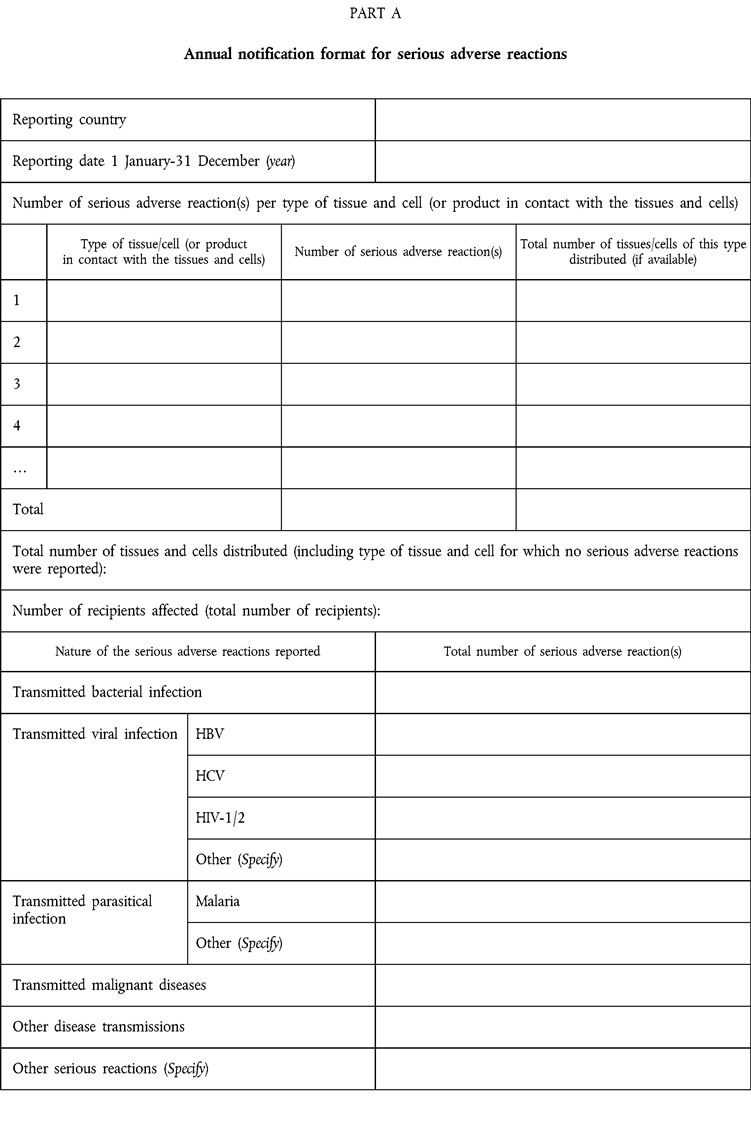
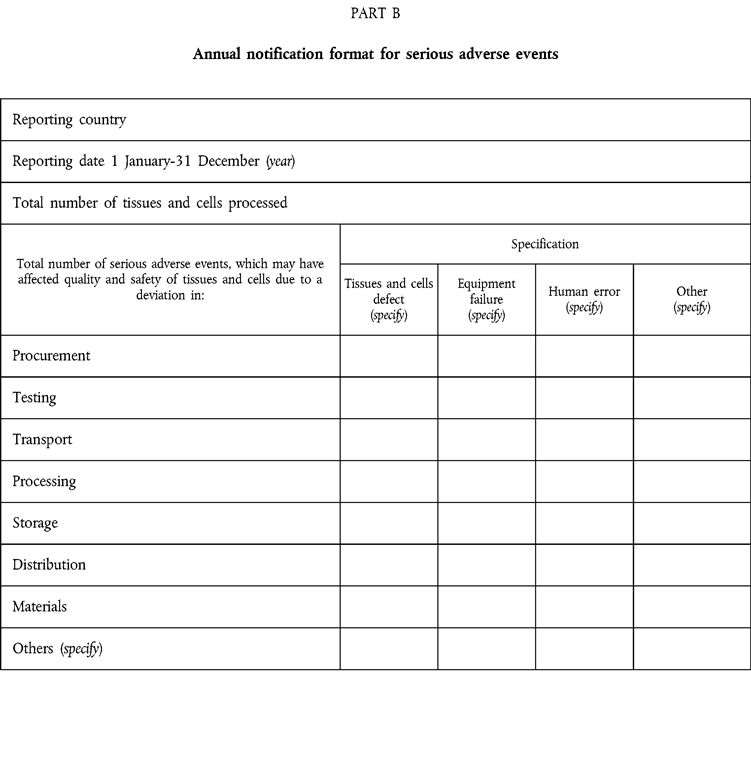
ANNEX VU.K.ANNUAL NOTIFICATION FORMAT
[F1ANNEX VI U.K. Minimum data to be kept in accordance with Article 9(2)
A. BY TISSUE ESTABLISHMENTS U.K.
(1) Donor identification U.K.
(2) Donation identification that will include at least: U.K.
Identification of the procurement organisation (including contact details) or the tissue establishment
Unique donation number
Date of procurement
Place of procurement
Type of donation (e.g. single v multi-tissue; autologous v allogenic; living v deceased)
(3) Product identification that will include at least: U.K.
Identification of the tissue establishment
Type of tissue and cell/product (basic nomenclature)
Pool number (in case of pooling)
Split number (if applicable)
Expiry date (if applicable)
Tissue/cell status (i.e. quarantined, suitable for use, etc.)
Description and origin of the products, processing steps applied, materials and additives coming into contact with tissues and cells and having an effect on their quality and/or safety.
Identification of the facility issuing the final label
(4) Single European Code (if applicable) U.K.
(5) Human application identification that will include at least: U.K.
Date of distribution/disposal
Identification of the clinician or end-user/facility
B. BY ORGANISATIONS RESPONSIBLE FOR HUMAN APPLICATION U.K.
(1) Identification of the supplier tissue establishment U.K.
(2) Identification of the clinician or end-user/facility U.K.
(3) Type of tissues and cells U.K.
(4) Product identification U.K.
(5) Identification of the recipient U.K.
(6) Date of application U.K.
(7) Single European Code (if applicable) U.K.
ANNEX VII U.K. THE STRUCTURE OF THE SINGLE EUROPEAN CODE
| DONATION IDENTIFICATION SEQUENCE | PRODUCT IDENTIFICATION SEQUENCE | |||||
| EU TISSUE ESTABLISHMENT CODE | UNIQUE DONATION NUMBER | PRODUCT CODE | SPLIT NUMBER | EXPIRY DATE (YYYYMMDD) | ||
| ISO country code | Tissue establishment number | Product Coding System identifier | Product number | |||
| 2 alphabetic characters | 6 alpha-numeric characters | 13 alpha-numeric characters | 1 alphabetic character | 7 alpha-numeric characters | 3 alpha-numeric characters | 8 numeric characters] |
[F2ANNEX VIII U.K. Data to be recorded in the EU Tissue Establishment Compendium
A. Tissue establishment information U.K.
1. Name of the tissue establishment U.K.
2. National or international code of tissue establishment U.K.
3. Name of the organisation in which the tissue establishment is located (if applicable) U.K.
4. Address of the tissue establishment U.K.
5. Publishable contact details: functional e-mail address, phone and fax U.K.
B. Details on the authorisation, accreditation, designation, or license of the tissue establishment U.K.
1. Name of the authorising, accrediting, designating or licensing competent authority or authorities U.K.
2. Name of the national competent authority or authorities responsible for maintenance of the EU Tissue Establishment Compendium U.K.
3. Name of the authorisation, accreditation, designation or licence holder (if applicable) U.K.
4. Tissues and cells for which the authorisation, accreditation, designation or license was granted U.K.
5. Activities actually carried out for which the authorisation, accreditation, designation or licence was granted U.K.
6. Status of the authorisation, accreditation, designation or license (authorised, suspended, revoked, in part or in full, voluntary cessation of activities) U.K.
7. Details of any conditions and exemptions added to the authorisation (if applicable).] U.K.
OJ L 169, 12.7.1993, p. 1. Directive as last amended by Regulation (EC) No 1882/2003 of the European Parliament and of the Council (OJ L 284, 31.10.2003, p. 1).
OJ L 331, 7.12.1998, p. 1. Directive as amended by Regulation (EC) No 1882/2003.
Options/Help
Print Options
PrintThe Whole Directive
PrintThe Annexes only
Legislation is available in different versions:
Latest Available (revised):The latest available updated version of the legislation incorporating changes made by subsequent legislation and applied by our editorial team. Changes we have not yet applied to the text, can be found in the ‘Changes to Legislation’ area.
Original (As adopted by EU): The original version of the legislation as it stood when it was first adopted in the EU. No changes have been applied to the text.
See additional information alongside the content
Geographical Extent: Indicates the geographical area that this provision applies to. For further information see ‘Frequently Asked Questions’.
Show Timeline of Changes: See how this legislation has or could change over time. Turning this feature on will show extra navigation options to go to these specific points in time. Return to the latest available version by using the controls above in the What Version box.
More Resources
Access essential accompanying documents and information for this legislation item from this tab. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as adopted version that was used for the EU Official Journal
- lists of changes made by and/or affecting this legislation item
- all formats of all associated documents
- correction slips
- links to related legislation and further information resources
Timeline of Changes
This timeline shows the different versions taken from EUR-Lex before exit day and during the implementation period as well as any subsequent versions created after the implementation period as a result of changes made by UK legislation.
The dates for the EU versions are taken from the document dates on EUR-Lex and may not always coincide with when the changes came into force for the document.
For any versions created after the implementation period as a result of changes made by UK legislation the date will coincide with the earliest date on which the change (e.g an insertion, a repeal or a substitution) that was applied came into force. For further information see our guide to revised legislation on Understanding Legislation.
More Resources
Use this menu to access essential accompanying documents and information for this legislation item. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as adopted version that was used for the print copy
- correction slips
Click 'View More' or select 'More Resources' tab for additional information including:
- lists of changes made by and/or affecting this legislation item
- confers power and blanket amendment details
- all formats of all associated documents
- links to related legislation and further information resources