- Latest available (Revised)
- Original (As adopted by EU)
Commission Implementing Regulation (EU) 2020/2235Show full title
Commission Implementing Regulation (EU) 2020/2235 of 16 December 2020 laying down rules for the application of Regulations (EU) 2016/429 and (EU) 2017/625 of the European Parliament and of the Council as regards model animal health certificates, model official certificates and model animal health/official certificates, for the entry into the Union and movements within the Union of consignments of certain categories of animals and goods, official certification regarding such certificates and repealing Regulation (EC) No 599/2004, Implementing Regulations (EU) No 636/2014 and (EU) 2019/628, Directive 98/68/EC and Decisions 2000/572/EC, 2003/779/EC and 2007/240/EC (Text with EEA relevance)
You are here:
- Show Geographical Extent(e.g. England, Wales, Scotland and Northern Ireland)
- Show Timeline of Changes
More Resources
When the UK left the EU, legislation.gov.uk published EU legislation that had been published by the EU up to IP completion day (31 December 2020 11.00 p.m.). On legislation.gov.uk, these items of legislation are kept up-to-date with any amendments made by the UK since then.
This item of legislation originated from the EU
Legislation.gov.uk publishes the UK version. EUR-Lex publishes the EU version. The EU Exit Web Archive holds a snapshot of EUR-Lex’s version from IP completion day (31 December 2020 11.00 p.m.).
Changes over time for: ANNEX III
Changes to legislation:
There are outstanding changes not yet made to Commission Implementing Regulation (EU) 2020/2235. Any changes that have already been made to the legislation appear in the content and are referenced with annotations.![]()
Changes to Legislation
Revised legislation carried on this site may not be fully up to date. Changes and effects are recorded by our editorial team in lists which can be found in the ‘Changes to Legislation’ area. Where those effects have yet to be applied to the text of the legislation by the editorial team they are also listed alongside the legislation in the affected provisions. Use the ‘more’ link to open the changes and effects relevant to the provision you are viewing.
Changes and effects yet to be applied to Annex III:
- Regulation incorporated (with modifications) by S.I. 2024/853 reg. 5 6 9 10(3)
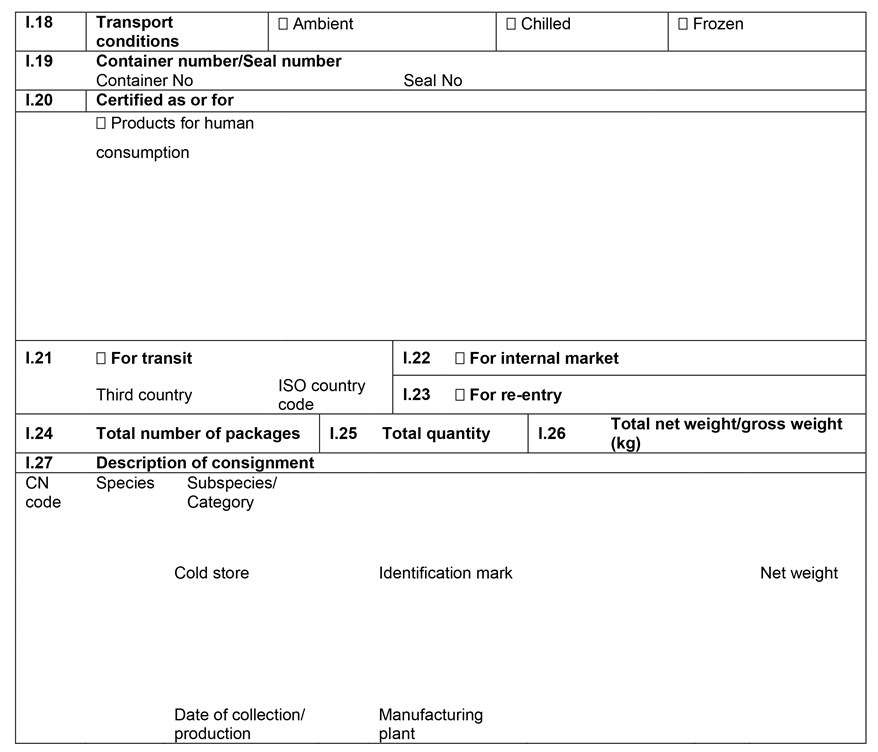
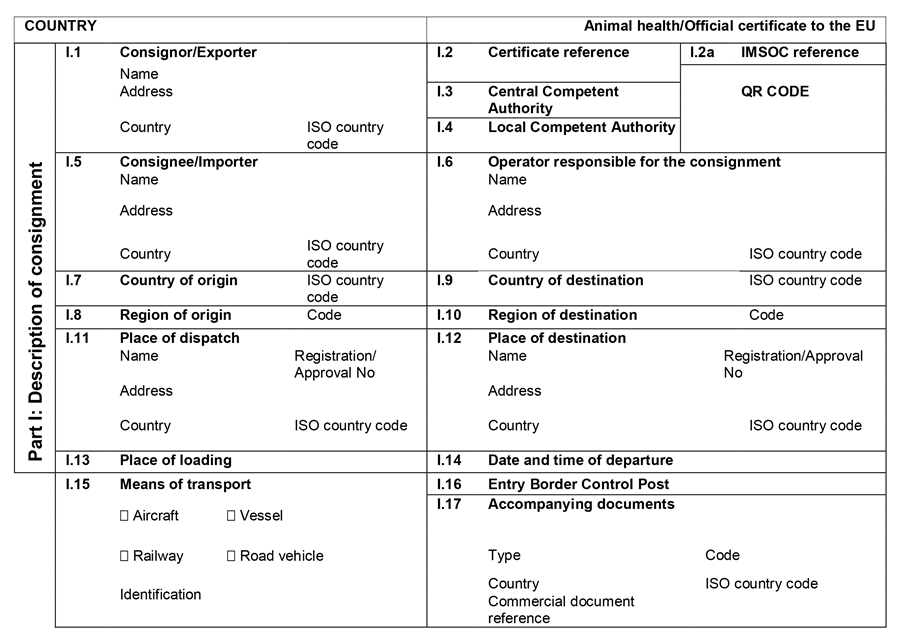
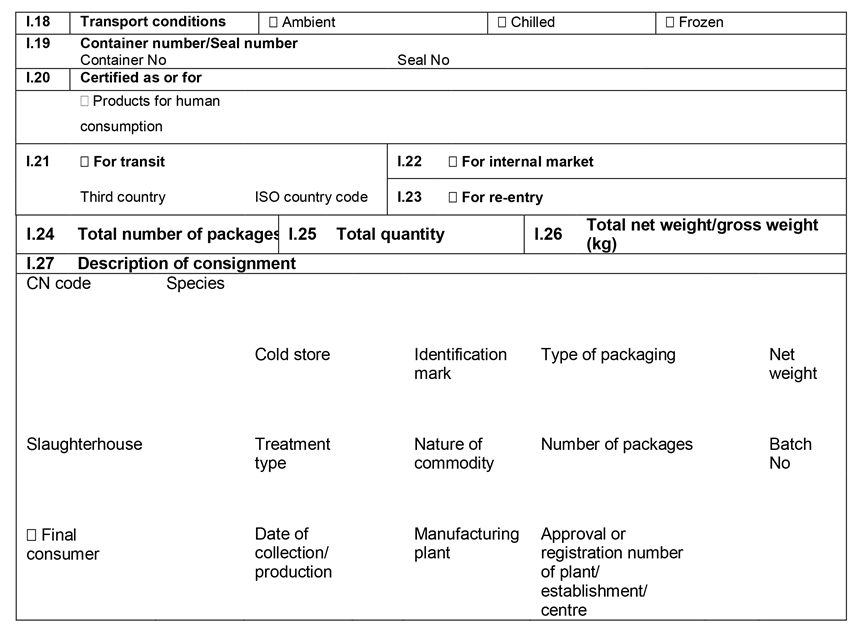
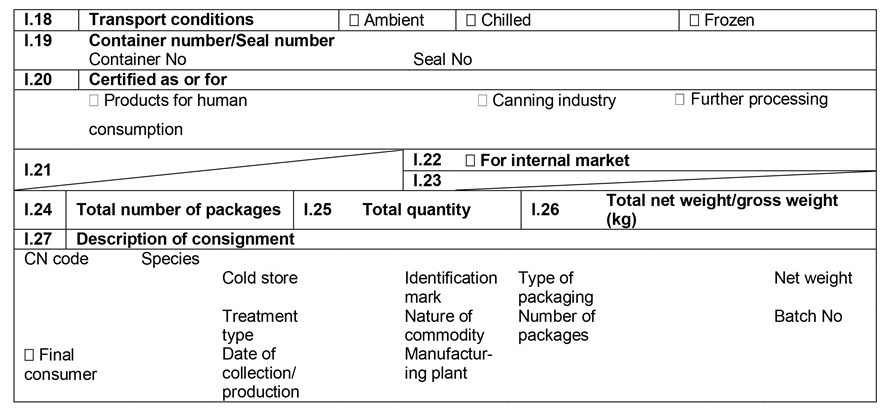
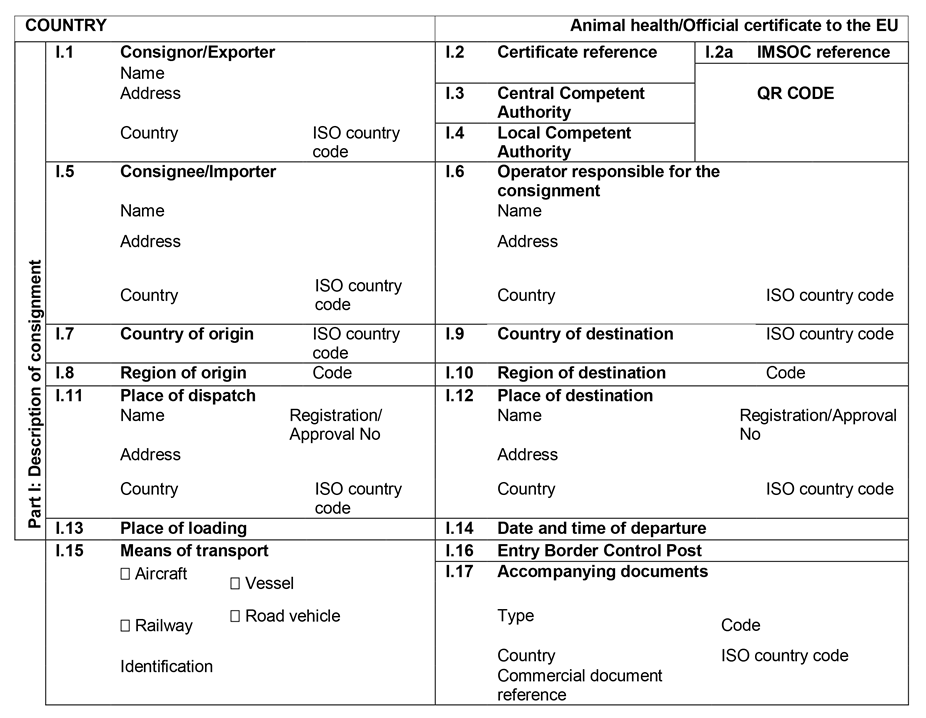
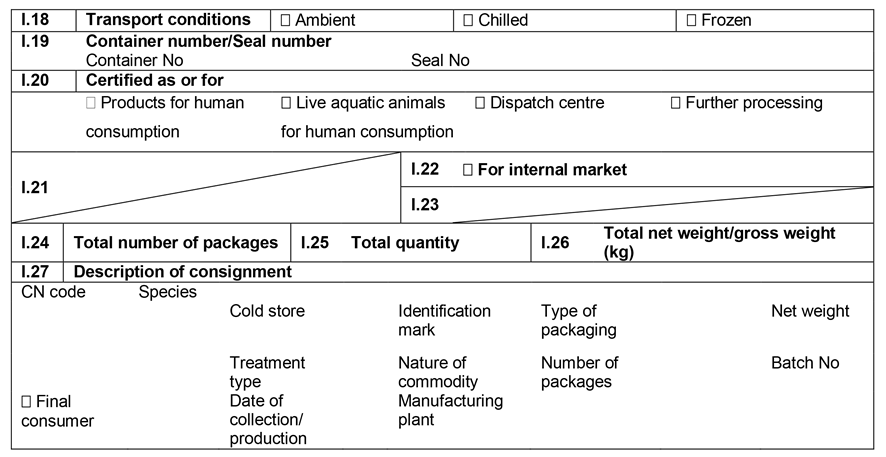
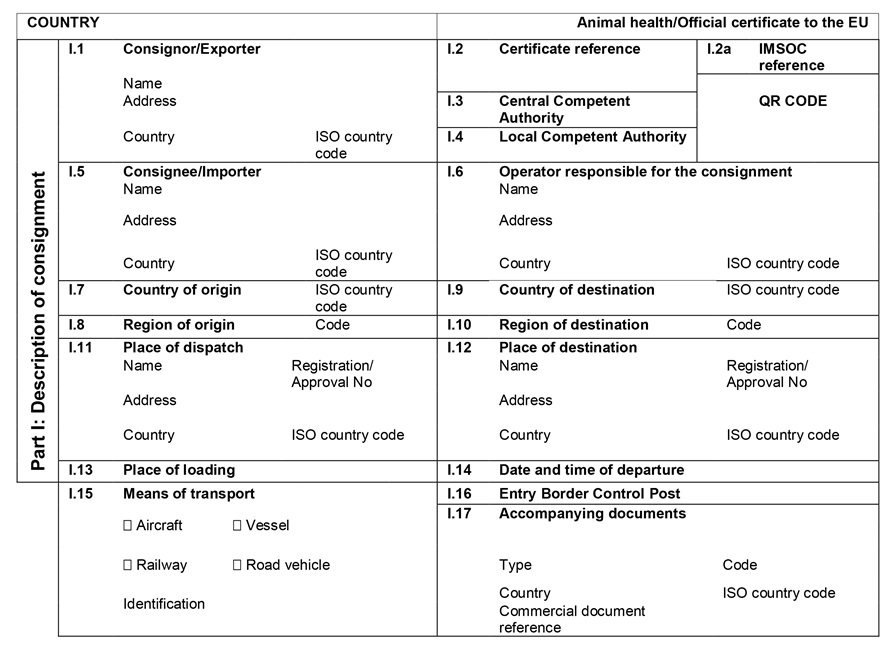
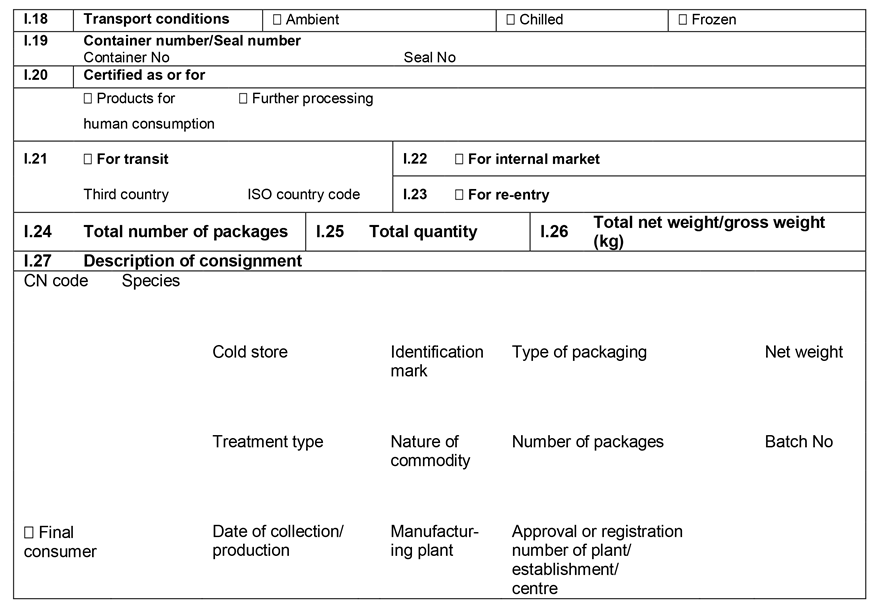
ANNEX IIIU.K.
Annex III contains the following model animal health/official certificates and official certificates for the entry into the Union:
MODELU.K.
| fresh meat of ungulates | |
| BOV | Chapter 1: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding mechanically separated meat, of domestic bovine animals |
| OVI | Chapter 2: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding mechanically separated meat, of domestic ovine and caprine animals |
| POR | Chapter 3: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding mechanically separated meat, of domestic porcine animals |
| EQU | Chapter 4: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding minced meat and mechanically separated meat, of domestic solipeds (Equus caballus, Equus asinus and their cross-breeds) |
| RUF | Chapter 5: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding offal, minced meat and mechanically separated meat, of animals of the family Bovidae (other than domestic bovine, ovine and caprine animals), camelid animals and cervid animals kept as farmed game |
| RUW | Chapter 6: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding offal, minced meat and mechanically separated meat, of wild animals of the family Bovidae (other than domestic bovine, ovine and caprine animals), wild camelid animals and wild cervid animals |
| SUF | Chapter 7: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding offal, minced meat and mechanically separated meat, of animals kept as farmed game of wild breeds of porcine animals and animals of the family Tayassuidae |
| SUW | Chapter 8: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding offal, minced meat and mechanically separated meat, of wild animals of wild breeds of porcine animals and animals of the family Tayassuidae |
| EQW | Chapter 9: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding offal, minced meat and mechanically separated meat, of wild game solipeds belonging to the subgenus Hippotigris (zebra) |
| RUM-MSM | Chapter 10: Model animal health/official certificate for the entry into the Union of mechanically separated meat, intended for human consumption, of domestic ruminants |
| SUI-MSM | Chapter 11: Model animal health/official certificate for the entry into the Union of mechanically separated meat, intended for human consumption, of domestic porcine animals |
| NZ-TRANSIT-SG | Chapter 12: Model animal health certificate for the entry into the Union of fresh meat intended for human consumption originating from New Zealand transiting through Singapore with unloading, possible storage and reloading before entry into the Union |
| meat of poultry, ratites and other game birds, eggs and egg products | |
| POU | Chapter 13: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding minced meat and mechanically separated meat, of poultry other than ratites |
| POU-MI/MSM | Chapter 14: Model animal health/official certificate for the entry into the Union of minced meat and mechanically separated meat, intended for human consumption, of poultry other than ratites |
| RAT | Chapter 15: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding minced meat and mechanically separated meat, of ratites |
| RAT-MI/MSM | Chapter 16: Model animal health/official certificate for the entry into the Union of minced meat and mechanically separated meat, intended for human consumption, of ratites |
| GBM | Chapter 17: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding minced meat and mechanically separated meat, of game birds |
| GBM-MI/MSM | Chapter 18: Model animal health/official certificate for the entry into the Union of minced meat and mechanically separated meat, intended for human consumption, of game-birds |
| E | Chapter 19: Model animal health/official certificate for the entry into the Union of eggs intended for human consumption |
| EP | Chapter 20: Model animal health/official certificate for the entry into the Union of egg products intended for human consumption |
| fresh meat, excluding mechanically separated meat, of wild leporidae, of certain wild land mammals and of farmed rabbits | |
| WL | Chapter 21: Model official certificate for the entry into the Union of fresh meat intended for human consumption of wild leporidae (rabbits and hares), excluding minced meat, mechanically separated meat and offal except for unskinned and uneviscerated leporidae |
| WM | Chapter 22: Model official certificate for the entry into the Union of fresh meat intended for human consumption, excluding offal, minced meat and mechanically separated meat, of wild land mammals other than ungulates and leporidae |
| RM | Chapter 23: Model animal health/official certificate for the entry into the Union of fresh meat intended for human consumption, excluding minced meat and mechanically separated meat, of farmed rabbits |
| meat preparations | |
| MP-PREP | Chapter 24: Model animal health/official certificate for the entry into the Union of meat preparations intended for human consumption |
| meat products, including rendered animal fats and greaves, meat extracts and treated stomachs, bladders, intestines others than casings | |
| MPNT | Chapter 25: Model animal health/official certificate for the entry into the Union of meat products intended for human consumption, including rendered animal fats and greaves, meat extracts and treated stomachs, bladders and intestines, others than casings, that are not required to undergo a specific risk-mitigating treatment |
| MPST | Chapter 26: Model animal health/official certificate for the entry into the Union of meat products intended for human consumption, including rendered animal fats and greaves, meat extracts and treated stomachs, bladders and intestines, others than casings, that are required to undergo a specific risk-mitigating treatment |
| casings | |
| CAS | Chapter 27: Model animal health/official certificate for the entry into the Union of casings intended for human consumption |
| live fish, live crustaceans and products of animal origin from those animals intended for human consumption | |
| FISH-CRUST-HC | Chapter 28: Model animal health/official certificate for the entry into the Union of live fish, live crustaceans and products of animal origin from those animals intended for human consumption |
| EU-FISH | Chapter 29: Model official certificate for the entry into the Union of fishery products intended for human consumption caught by vessels flying the flag of a Member State and transferred in third countries with or without storage |
| FISH/MOL-CAP | Chapter 30: Model official certificate for the entry into the Union of fishery products or fishery products derived from bivalve molluscs intended for human consumption entering the Union directly from a reefer, freezer or factory vessel flying the flag of a third country as provided for in Article 11(3) of Delegated Regulation (EU) 2019/625 |
| live bivalve molluscs, echinoderms, tunicates, marine gastropods and products of animal origin from those animals | |
| MOL-HC | Chapter 31: Model animal health/official certificate for the entry into the Union of live bivalve molluscs, echinoderms, tunicates, marine gastropods and products of animal origin from those animals intended for human consumption |
| MOL-AT | Chapter 32: Model official certificate for the entry into the Union of processed bivalve molluscs intended for human consumption belonging to the species Acanthocardia Tuberculatum |
| raw milk, dairy products, colostrum, and colostrum-based products | |
| MILK-RM | Chapter 33: Model animal health/official certificate for the entry into the Union of raw milk intended for human consumption |
| MILK-RMP/NT | Chapter 34: Model animal health/official certificate for the entry into the Union of dairy products intended for human consumption derived from raw milk or that are not required to undergo a specific risk-mitigating treatment |
| DAIRY-PRODUCTS-PT | Chapter 35: Model animal health/official certificate for the entry into the Union of dairy products intended for human consumption that are required to undergo a pasteurization treatment |
| DAIRY-PRODUCTS-ST | Chapter 36: Model animal health/official certificate for the entry into the Union of dairy products intended for human consumption that are required to undergo a specific risk-mitigating treatment other than pasteurization |
| COLOSTRUM | Chapter 37: Model animal health/official certificate for the entry into the Union of colostrum intended for human consumption |
| COLOSTRUM-BP | Chapter 38: Model animal health/official certificate for the entry into the Union of colostrum-based products intended for human consumption |
| chilled, frozen or prepared frogs’ legs | |
| FRG | Chapter 39: Model official certificate for the entry into the Union of chilled, frozen or prepared frogs’ legs intended for human consumption |
| snails | |
| SNS | Chapter 40: Model official certificate for the entry into the Union of snails intended for human consumption |
| gelatine | |
| GEL | Chapter 41: Model official certificate for the entry into the Union of gelatine intended for human consumption |
| collagen | |
| COL | Chapter 42: Model official certificate for the entry into the Union of collagen intended for human consumption |
| raw materials for the production of gelatine and collagen | |
| RCG | Chapter 43: Model animal health/official certificate for the entry into the Union of raw materials for the production of gelatine and collagen intended for human consumption |
| treated raw materials for the production of gelatine and collagen | |
| TCG | Chapter 44: Model animal health/official certificate for the entry into the Union of treated raw materials for the production of gelatine and collagen intended for human consumption |
| honey and other apiculture products intended for human consumption | |
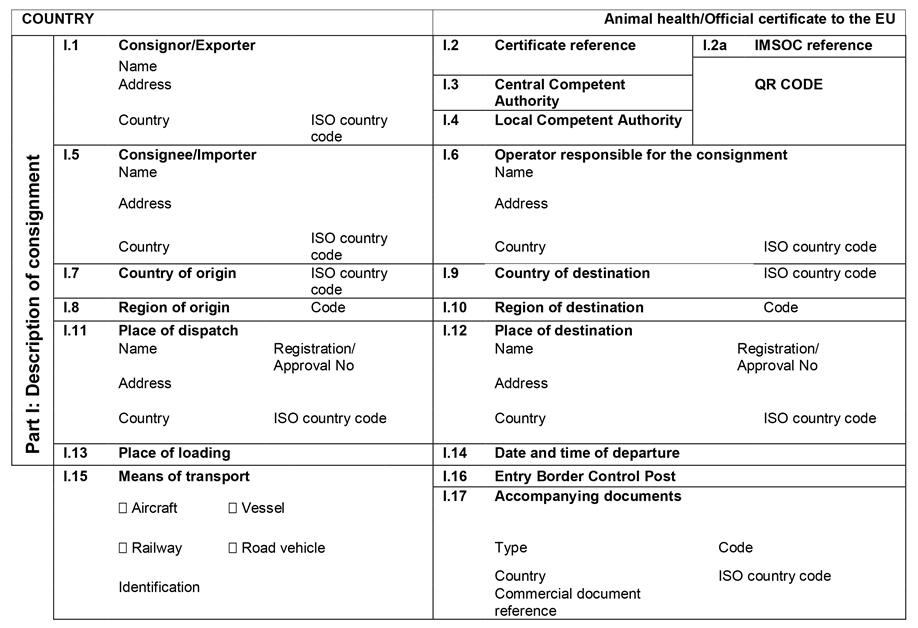
| HON | Chapter 45: Model official certificate for the entry into the Union of honey and other apiculture products intended for human consumption |
| highly refined chondroitin sulphate, hyaluronic acid, other hydrolysed cartilage products, chitosan, glucosamine, rennet, isinglass and amino acids | |
| HRP | Chapter 46: Model official certificate for the entry into the Union of highly refined chondroitin sulphate, hyaluronic acid, other hydrolysed cartilage products, chitosan, glucosamine, rennet, isinglass and amino acids intended for human consumption |
| reptile meat | |
| REP | Chapter 47: Model official certificate for the entry into the Union of reptile meat intended for human consumption |
| insects | |
| INS | Chapter 48: Model official certificate for the entry into the Union of insects intended for human consumption |
| other products of animal origin | |
| PAO | Chapter 49: Model official certificate for the entry into the Union of other products of animal origin derived from domestic ungulates, poultry, rabbits or fishery products intended for human consumption and not covered by Articles 8 to 26 of Commission Implementing Regulation (EU) 2020/2235 |
| composite products | |
| COMP | Chapter 50: Model animal health/official certificate for the entry into the Union of not shelf-stable composite products and shelf-stable composite products, containing any quantity of meat products except gelatine, collagen and highly refined products, and intended for human consumption |
| sprouts intended for human consumption and seeds intended for the production of sprouts for human consumption | |
| SPR | Chapter 51: Model official certificate for the entry into the Union of sprouts intended for human consumption and seeds intended for the production of sprouts for human consumption |
| transit through the Union to a third country either by immediate transit or after storage in the Union of composite products | |
| TRANSIT-COMP | Chapter 52: Model animal health certificate for the transit through the Union to a third country either by immediate transit or after storage in the Union of not shelf-stable composite products and shelf-stable composite products containing any quantity of meat products and intended for human consumption |
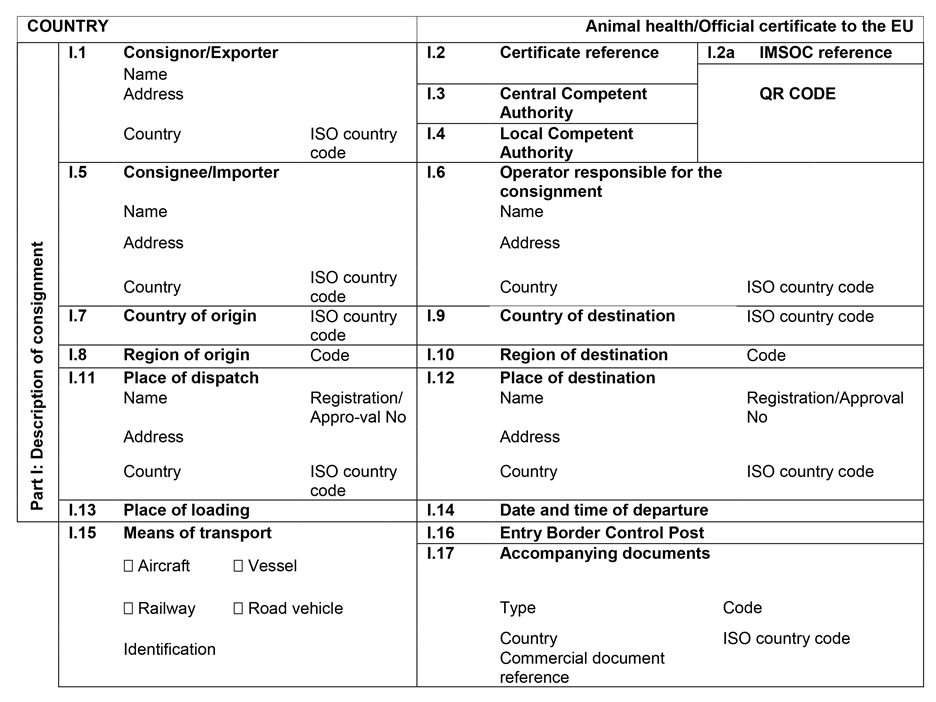
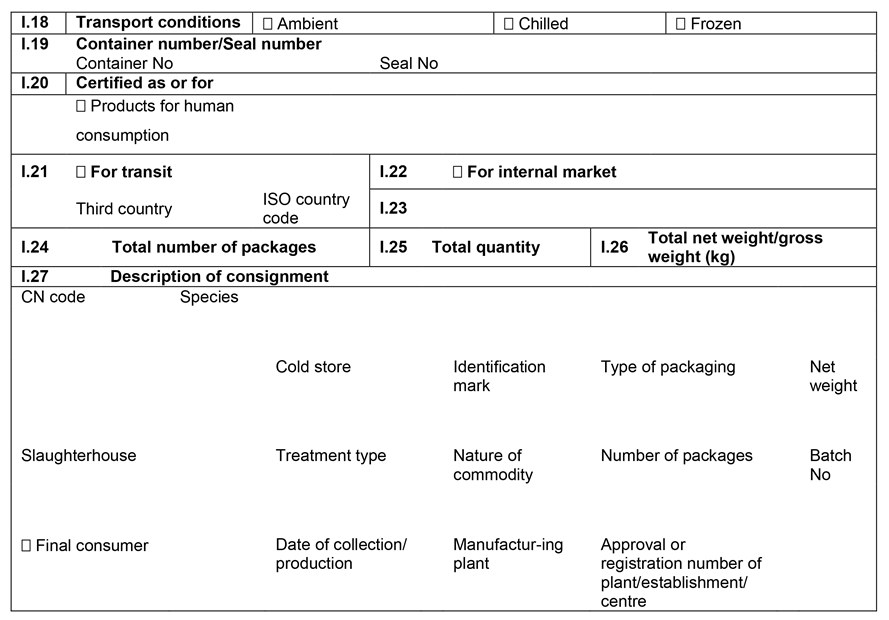
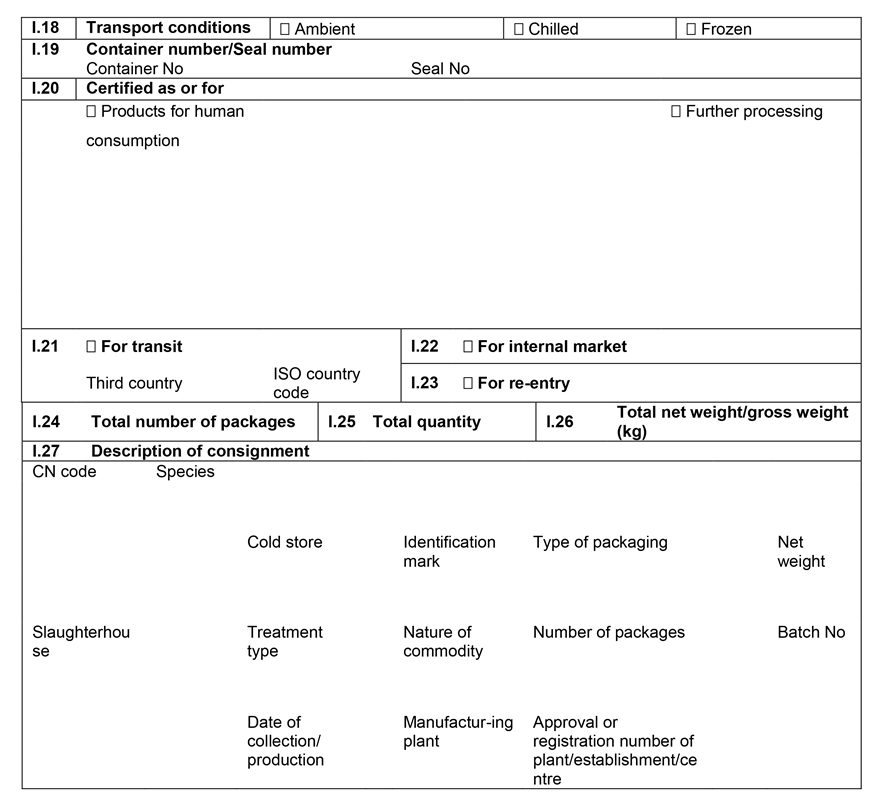
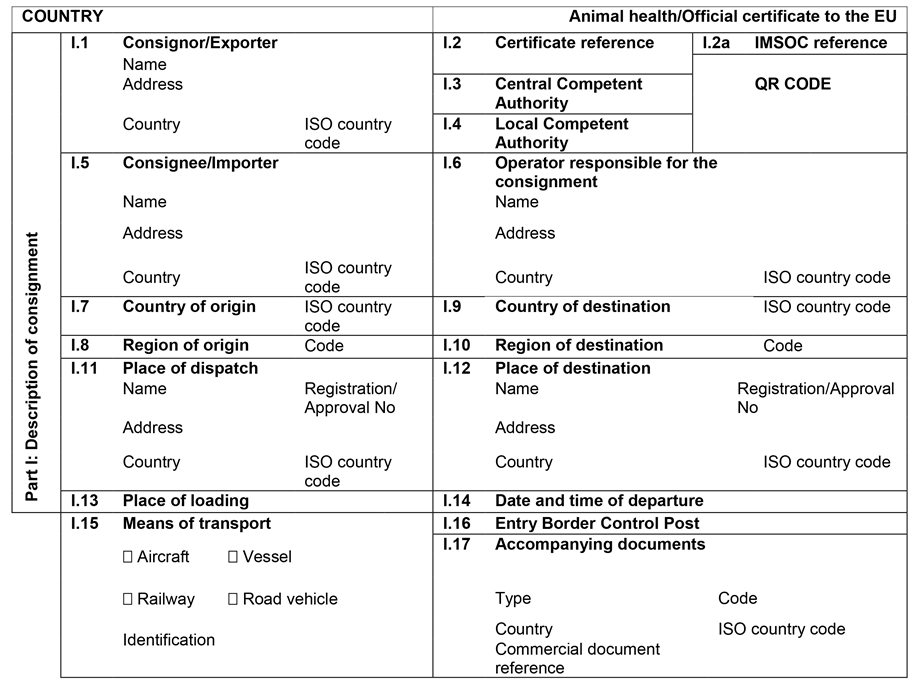
CHAPTER 1U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MECHANICALLY SEPARATED MEAT, OF DOMESTIC BOVINE ANIMALS (MODEL BOV)
CHAPTER 2U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MECHANICALLY SEPARATED MEAT, OF DOMESTIC OVINE AND CAPRINE ANIMALS (MODEL OVI)
CHAPTER 3U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MECHANICALLY SEPARATED MEAT, OF DOMESTIC PORCINE ANIMALS (MODEL POR)
CHAPTER 4U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF DOMESTIC SOLIPEDS (EQUUS CABALLUS, EQUUS ASINUS AND THEIR CROSS-BREEDS) (MODEL EQU)
CHAPTER 5U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING OFFAL, MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF ANIMALS OF THE FAMILY BOVIDAE (OTHER THAN DOMESTIC BOVINE, OVINE AND CAPRINE ANIMALS), CAMELID ANIMALS AND CERVID ANIMALS KEPT AS FARMED GAME (MODEL RUF)
CHAPTER 6U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING OFFAL, MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF WILD ANIMALS OF THE FAMILY BOVIDAE (OTHER THAN DOMESTIC BOVINE, OVINE AND CAPRINE ANIMALS), WILD CAMELID ANIMALS AND WILD CERVID ANIMALS (MODEL RUW)
CHAPTER 7U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING OFFAL, MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF ANIMALS KEPT AS FARMED GAME OF WILD BREEDS OF PORCINE ANIMALS AND ANIMALS OF THE FAMILY TAYASSUIDAE (MODEL SUF)
CHAPTER 8U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING OFFAL, MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF WILD ANIMALS OF WILD BREEDS OF PORCINE ANIMALS AND ANIMALS OF THE FAMILY TAYASSUIDAE (MODEL SUW)
CHAPTER 9U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING OFFAL, MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF WILD GAME SOLIPEDS BELONGING TO THE SUBGENUS HIPPOTIGRIS (ZEBRA) (MODEL EQW)
CHAPTER 10U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MECHANICALLY SEPARATED MEAT, INTENDED FOR HUMAN CONSUMPTION, OF DOMESTIC RUMINANTS (MODEL RUM-MSM)
CHAPTER 11U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MECHANICALLY SEPARATED MEAT, INTENDED FOR HUMAN CONSUMPTION, OF DOMESTIC PORCINE ANIMALS (MODEL SUI-MSM)
CHAPTER 12U.K. MODEL ANIMAL HEALTH CERTIFICATE FOR THE ENTRY IN TO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION ORIGINATING FROM NEW ZEALAND TRANSITING THROUGH SINGAPORE WITH UNLOADING, POSSIBLE STORAGE AND RELOADING BEFORE ENTRY INTO THE UNION (MODEL NZ-TRANSIT-SG)
CHAPTER 13U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF POULTRY OTHER THAN RATITES (MODEL POU)
CHAPTER 14U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MINCED MEAT AND MECHANICALLY SEPARATED MEAT, INTENDED FOR HUMAN CONSUMPTION, OF POULTRY OTHER THAN RATITES (MODEL POU-MI/MSM)
NOT AVAILABLE YET
CHAPTER 15U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF RATITES (MODEL RAT)
CHAPTER 16U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MINCED MEAT AND MECHANICALLY SEPARATED MEAT, INTENDED FOR HUMAN CONSUMPTION, OF RATITES (MODEL RAT-MI/MSM)
NOT AVAILABLE YET
CHAPTER 17U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF GAME BIRDS (MODEL GBM)
CHAPTER 18U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MINCED MEAT AND MECHANICALLY SEPARATED MEAT, INTENDED FOR HUMAN CONSUMPTION, OF GAME-BIRDS (MODEL GBM-MI/MSM)
NOT AVAILABLE YET
CHAPTER 19U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF EGGS INTENDED FOR HUMAN CONSUMPTION (MODEL E)
CHAPTER 20U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF EGG PRODUCTS INTENDED FOR HUMAN CONSUMPTION (MODEL EP)
CHAPTER 21U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION OF WILD LEPORIDAE (RABBITS AND HARES), EXCLUDING MINCED MEAT, MECHANICALLY SEPARATED MEAT AND OFFAL EXCEPT FOR UNSKINNED AND UNEVISCERATED LEPORIDAE (MODEL WL)
CHAPTER 22U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING OFFAL, MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF WILD LAND MAMMALS OTHER THAN UNGULATES AND LEPORIDAE (MODEL WM)
CHAPTER 23U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FRESH MEAT INTENDED FOR HUMAN CONSUMPTION, EXCLUDING MINCED MEAT AND MECHANICALLY SEPARATED MEAT, OF FARMED RABBITS (MODEL RM)
CHAPTER 24U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MEAT PREPARATIONS INTENDED FOR HUMAN CONSUMPTION (MODEL MP-PREP)
CHAPTER 25U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MEAT PRODUCTS INTENDED FOR HUMAN CONSUMPTION, INCLUDING RENDERED ANIMAL FATS AND GREAVES, MEAT EXTRACTS AND TREATED STOMACHS, BLADDERS AND INTESTINES OTHERS THAN CASINGS, THAT ARE NOT REQUIRED TO UNDERGO A SPECIFIC RISK-MITIGATING TREATMENT (MODEL MPNT)
CHAPTER 26U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF MEAT PRODUCTS INTENDED FOR HUMAN CONSUMPTION, INCLUDING RENDERED ANIMAL FATS AND GREAVES, MEAT EXTRACTS AND TREATED STOMACHS, BLADDERS AND INTESTINES, OTHERS THAN CASINGS, THAT ARE REQUIRED TO UNDERGO A SPECIFIC RISK-MITIGATING TREATMENT (MODEL MPST)
CHAPTER 27U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF CASINGS INTENDED FOR HUMAN CONSUMPTION (MODEL CAS)
CHAPTER 28U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY IN THE UNION OF LIVE FISH, LIVE CRUSTACEANS AND PRODUCTS OF ANIMAL ORIGIN FROM THOSE ANIMALS INTENDED FOR HUMAN CONSUMPTION (MODEL FISH-CRUST-HC)
CHAPTER 29U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FISHERY PRODUCTS INTENDED FOR HUMAN CONSUMPTION CAUGHT BY VESSELS FLYING THE FLAG OF A MEMBER STATE AND TRANSFERRED IN THIRD COUNTRIES WITH OR WITHOUT STORAGE (MODEL EU-FISH)
CHAPTER 30U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF FISHERY PRODUCTS OR FISHERY PRODUCTS DERIVED FROM BIVALVE MOLLUSCS INTENDED FOR HUMAN CONSUMPTION ENTERING THE UNION DIRECTLY FROM A REEFER, FREEZEROR FACTORY VESSEL FLYING THE FLAG OF A THIRD COUNTRY AS PROVIDED FOR IN ARTICLE 11(3) OF DELEGATED REGULATION (EU) 2019/625 (MODEL FISH/MOL-CAP)
CHAPTER 31U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY IN THE UNION OF LIVE BIVALVE MOLLUSCS, ECHINODERMS, TUNICATES, MARINE GASTROPODS AND PRODUCTS OF ANIMAL ORIGIN FROM THESE ANIMALS INTENDED FOR HUMAN CONSUMPTION (MODEL MOL-HC)
CHAPTER 32U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF PROCESSED BIVALVE MOLLUSCS INTENDED FOR HUMAN CONSUMPTION BELONGING TO THE SPECIES ACANTHOCARDIA TUBERCULATUM (MODEL MOL-AT)
The certifying officer hereby certifies that the processed bivalve molluscs of the species Acanthocardia tuberculatum, certified in the official certificate reference No:…
(1)were harvested in production areas clearly identified, classified and monitored by the competent authorities in accordance with Articles 52 and 59 of Commission Implementing Regulation (EU) 2019/627(1) and where the paralytic shellfish poisoning (PSP) toxin quantity is lower than 300 μg for 100g;U.K.
(2)were transported in containers or vehicles sealed by the competent authority, directly to the establishment:U.K.
……
……
(name and official approval number of the establishment, authorised specially by the competent authorities to carry out their treatment);
(3)were accompanied while being transported to this establishment by a document issued by the competent authorities which authorise the transport, attesting to the nature and quantity of the product, production area of origin and establishment of destination;U.K.
(4)were subjected to the heat treatment outlined in the Annex to Commission Decision 96/77/EC(2); andU.K.
(5)after heat treatment they do not contain PSP toxins quantity that exceeds 80 μg for 100g using an Union official method, as demonstrated by the attached analytical report(s) of the test carried out on each lot included in the consignment covered by this certificate.U.K.
The certifying officer hereby certifies that the competent authorities have verified that the ‘own’ checks carried out in the establishment referred to in point (2) are specifically applied to the heat treatment referred to in point 4.
The undersigned certifying officer hereby declares that he/she is aware of the requirements of Decision 96/77/EC and that the attached analytical report(s) correspond(s) to the test carried out on the products after processing.
| Certifying officer | |
| Name (in capital letters) | |
| Date | Qualification and title |
| Stamp | Signature |
CHAPTER 33U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF RAW MILK INTENDED FOR HUMAN CONSUMPTION (MODEL MILK-RM)
CHAPTER 34U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF DAIRY PRODUCTS INTENDED FOR HUMAN CONSUMPTION DERIVED FROM RAW MILK OR THAT ARE NOT REQUIRED TO UNDERGO A SPECIFIC RISK-MITIGATING TREATMENT (MODEL MILK-RMP/NT)
CHAPTER 35U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF DAIRY PRODUCTS INTENDED FOR HUMAN CONSUMPTION THAT ARE REQUIRED TO UNDERGO A PASTEURIZATION TREATMENT (MODEL DAIRY-PRODUCTS-PT)
CHAPTER 36U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF DAIRY PRODUCTS INTENDED FOR HUMAN CONSUMPTION THAT ARE REQUIRED TO UNDERGO A SPECIFIC RISK-MITIGATING TREATMENT OTHER THAN PASTEURIZATION (MODEL DAIRY-PRODUCTS-ST)
CHAPTER 37U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF COLOSTRUM INTENDED FOR HUMAN CONSUMPTION (MODEL COLOSTRUM)
CHAPTER 38U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF COLOSTRUM-BASED PRODUCTS INTENDED FOR HUMAN CONSUMPTION (MODEL COLOSTRUM-BP)
CHAPTER 39U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF CHILLED, FROZEN OR PREPARED FROGS’ LEGS INTENDED FOR HUMAN CONSUMPTION (MODEL FRG)
CHAPTER 40U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF SNAILS INTENDED FOR HUMAN CONSUMPTION (MODEL SNS)
CHAPTER 41U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF GELATINE INTENDED FOR HUMAN CONSUMPTION (MODEL GEL)
CHAPTER 42U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF COLLAGEN INTENDED FOR HUMAN CONSUMPTION (MODEL COL)
CHAPTER 43U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF RAW MATERIALS FOR THE PRODUCTION OF GELATINE AND COLLAGEN INTENDED FOR HUMAN CONSUMPTION (MODEL RCG)
CHAPTER 44U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF TREATED RAW MATERIALS FOR THE PRODUCTION OF GELATINE AND COLLAGEN INTENDED FOR HUMAN CONSUMPTION (MODEL TCG)
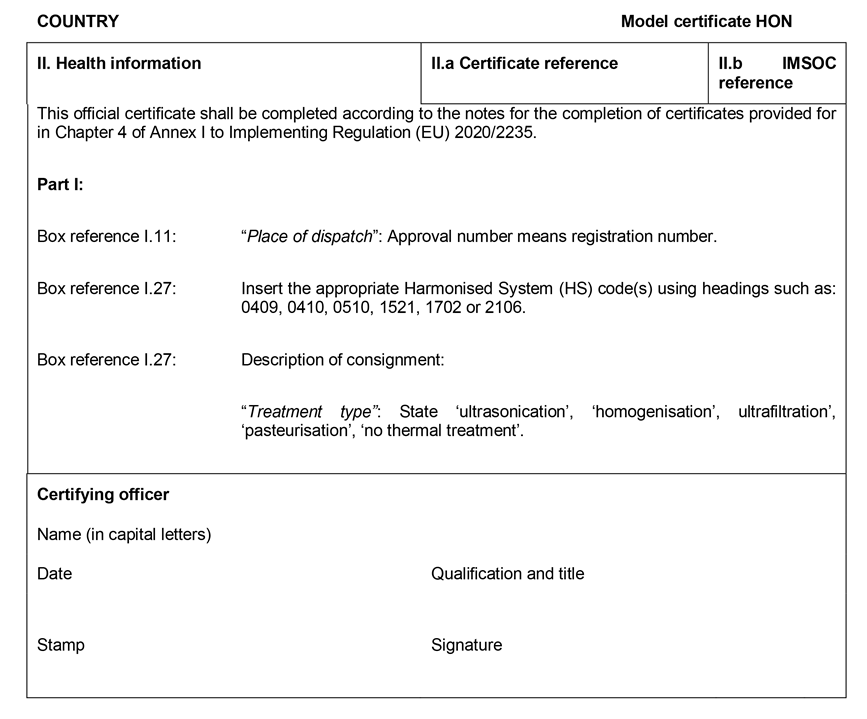
CHAPTER 45U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF HONEY AND OTHER APICULTURE PRODUCTS INTENDED FOR HUMAN CONSUMPTION (MODEL HON)
CHAPTER 46U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF HIGHLY REFINED CHONDROITIN SULPHATE, HYALURONIC ACID, OTHER HYDROLYSED CARTILAGE PRODUCTS, CHITOSAN, GLUCOSAMINE, RENNET, ISINGLASS AND AMINO ACIDS INTENDED FOR HUMAN CONSUMPTION (MODEL HRP)
CHAPTER 47U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF REPTILE MEAT INTENDED FOR HUMAN CONSUMPTION (MODEL REP)
CHAPTER 48U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF INSECTS INTENDED FOR HUMAN CONSUMPTION (MODEL INS)
CHAPTER 49U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF OTHER PRODUCTS OF ANIMAL ORIGIN DERIVED FROM DOMESTIC UNGULATES, POULTRY, RABBITS OR FISHERY PRODUCTS INTENDED FOR HUMAN CONSUMPTION AND NOT COVERED BY ARTICLES 8 TO 26 OF COMMISSION IMPLEMENTING REGULATION (EU) 2020/2235 (MODEL PAO)
CHAPTER 50U.K. MODEL ANIMAL HEALTH/OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF NOT SHELF-STABLE COMPOSITE PRODUCTS AND SHELF-STABLE COMPOSITE PRODUCTS, CONTAINING ANY QUANTITY OF MEAT PRODUCTS EXCEPT GELATINE, COLLAGEN AND HIGHLY REFINED PRODUCTS, AND INTENDED FOR HUMAN CONSUMPTION (MODEL COMP)
CHAPTER 51U.K. MODEL OFFICIAL CERTIFICATE FOR THE ENTRY INTO THE UNION OF SPROUTS INTENDED FOR HUMAN CONSUMTION AND SEEDS INTENDED FOR THE PRODUCTION OF SPROUTS FOR HUMAN CONSUMPTION (MODEL SPR)
CHAPTER 52U.K. MODEL ANIMAL HEALTHCERTIFICATE FOR THE TRANSIT THROUGH THE UNION TO A THIRD COUNTRY EITHER BY IMMEDIATE TRANSIT OR AFTER STORAGE IN THE UNION OF NOT SHELF-STABLE COMPOSITE PRODUCTS AND SHELF-STABLE COMPOSITE PRODUCTS CONTAINING ANY QUANTITY OF MEAT PRODUCTS AND INTENDED FOR HUMAN CONSUMPTION (MODEL TRANSIT-COMP)
Commission Implementing Regulation (EU) 2019/627 of 15 March 2019 laying down uniform practical arrangements for the performance of official controls on products of animal origin intended for human consumption in accordance with Regulation (EU) 2017/625 of the European Parliament and of the Council and amending Commission Regulation (EC) No 2074/2005 as regards official controls (OJ L 131, 17.5.2019, p. 51).
Commission Decision 96/77/EC of 18 January 1996 establishing the conditions for the harvesting and processing of certain bivalve molluscs coming from areas where the paralytic shellfish poison level exceeds the limit laid down by Council Directive 91/492/EEC (OJ L 15, 20.1.1996, p. 46).
Options/Help
Print Options
PrintThe Whole Regulation
PrintThis Annex only
Legislation is available in different versions:
Latest Available (revised):The latest available updated version of the legislation incorporating changes made by subsequent legislation and applied by our editorial team. Changes we have not yet applied to the text, can be found in the ‘Changes to Legislation’ area.
Original (As adopted by EU): The original version of the legislation as it stood when it was first adopted in the EU. No changes have been applied to the text.
See additional information alongside the content
Geographical Extent: Indicates the geographical area that this provision applies to. For further information see ‘Frequently Asked Questions’.
Show Timeline of Changes: See how this legislation has or could change over time. Turning this feature on will show extra navigation options to go to these specific points in time. Return to the latest available version by using the controls above in the What Version box.
More Resources
Access essential accompanying documents and information for this legislation item from this tab. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as adopted version that was used for the EU Official Journal
- lists of changes made by and/or affecting this legislation item
- all formats of all associated documents
- correction slips
- links to related legislation and further information resources
Timeline of Changes
This timeline shows the different versions taken from EUR-Lex before exit day and during the implementation period as well as any subsequent versions created after the implementation period as a result of changes made by UK legislation.
The dates for the EU versions are taken from the document dates on EUR-Lex and may not always coincide with when the changes came into force for the document.
For any versions created after the implementation period as a result of changes made by UK legislation the date will coincide with the earliest date on which the change (e.g an insertion, a repeal or a substitution) that was applied came into force. For further information see our guide to revised legislation on Understanding Legislation.
More Resources
Use this menu to access essential accompanying documents and information for this legislation item. Dependent on the legislation item being viewed this may include:
- the original print PDF of the as adopted version that was used for the print copy
- correction slips
Click 'View More' or select 'More Resources' tab for additional information including:
- lists of changes made by and/or affecting this legislation item
- confers power and blanket amendment details
- all formats of all associated documents
- links to related legislation and further information resources